Biotechnology Development in Cuba: Challenges in the Economic Strategy Beyond 2021
Despite great advances and results, Cuba’s biotechnology sector faces major challenges in terms of the concentration of its export destinations and a lack of financial resources.
Since 1959, a central objective of the Cuban government has been the country’s development from a national science standpoint; to that end, it established multiple educational programs and granted important budgetary allocations toward the formation of the necessary professional labor force.
Among the Revolutionary government’s first steps were the literacy campaign (1961), the reactivation of the Cuban Academy of Sciences (1962), and the creation of several Public Health institutes. Beginning in 1962, Cuba carried out a nationwide immunization program and introduced oral polio vaccination, making it the first country to eliminate the disease.
During the 1980s, a development strategy for biotechnology was proposed along with the creation, in 1981, of several leading-edge scientific institutions. Thereafter, other biological institutions were established, including:
• The Center for Biological Research, the Center for Genetic Engineering and Biotechnology (CIGB)
• The Center for the Production of Laboratory Animals (CENPALAB)
• The National Center for Biopreparations (BIOCEN)
• The Center for Molecular Immunology (CIM)
• The Center for Pharmaceutical Chemistry
Other centers were also built and equipment was installed in several universities and provinces. In 1965, the National Center for Scientific Research (CNIC) was inaugurated.
Despite the deep economic and social crisis that Cuba went through from 1990–1996, known as the Special Period, the State’s budgetary investments in the high-tech realm continued and large sums were allocated for equipment.
In 1992, the Scientific Pole (a scientific park in western Havana) was established, comprising more than fifty institutions and 10,000 workers. This structure allowed all the member companies to have their own commercial company to facilitate the export process; that is, they operated in a closed cycle.
From the beginning, this strategy was based on having highly qualified personnel who participated in the “closed cycle” (research + development + production + marketing), wherein the effects of specific results could also be measured, based on how many diseases were avoided and how many people were cured. Researchers were also offered job incentives, good working conditions, and the possibility of ongoing training in Cuba and abroad, although less so today.
The abundance and low cost of a highly qualified labor force relative to more developed countries meant that Cuban products had proportionally lower research and production costs.
Evolution of Results
The experience of Cuban biotechnology can be considered a success when measured by certain criteria: product development (biopharmaceuticals and vaccines), impact on public health, registration of patents, and exports.
These results are thanks to its prioritization and the large investments in these areas, but it has been difficult to determine whether the total investment costs have been recovered over time.
In general, the Cuban industry has lacked autonomy in making financial decisions. This often makes it impossible to obtain capital when it is needed, and therefore the lack of financial resources is one of its main weaknesses.
Despite the numerous applications of biotechnology in various sectors, given Cuba’s limited resources, biotechnological products have mainly been concentrated in three sectors: human health, research on livestock, and agriculture.
The social impact of national biotechnological products can be seen in the various vaccines developed and in the overall impact on the population, such as the cases of meningitis B/C, hepatitis B, and dengue; technologies for the diagnosis of neural tube defects; pregnancy kits; lung cancer vaccines; and drugs to combat viral diseases, myocardial infarction, and organ transplant rejection, among others.
Bivalent, trivalent, quadrivalent and pentavalent vaccines have been achieved, as well as other products to combat AIDS, which delay the onset of the disease in already infected people and in preventive variants.
A new therapeutic vaccine was recently developed for chronic hepatitis B: HeberNasvac. It has been shown to have higher levels of efficacy than the primary registered treatments for this condition. It was one of the first vaccines in the world for therapeutic use and it is applied nasally and subcutaneously. It is the first to contain two antigens of the hepatitis B virus—the surface antigen and the nucleocapsid.

The Center for Genetic Engineering and Biotechnology (CIGB) developed one of Cuba’s main biotechnology export products: Heberprot-P, the only drug in the world to reduce the amputation rate in patients with diabetic foot ulcers by up to 75%.
The interesting thing about Cuban biotechnology is its specialization in preventive vaccines produced locally. Among them, the following stand out:
• Hepatitis B vaccine. Created by the CIGB.
• BC meningococcal vaccine. Created by the company The Finlay Institute.
• Rabies vaccine. Created by The Finlay Institute.
• Tetanus-toxoid vaccine. Created by The Finlay Institute.
• Diphtheria-tetanus vaccine. Created by The Finlay Institute.
• Diphtheria, Tetanus, and Pertussis (DTP) vaccine. Created by The Finlay Institute.
• Trivalent leptospirosis vaccine. Created by The Finlay Institute.
• DTP-HB quadrivalent vaccine (TRivac-Hb). Created by the CIGB.
• Heberpenta pentavalent vaccine. Created by the CIGB.
• Typhoid vaccine. Created by The Finlay Institute.
• Vaccine against Haemophilus Influenza type B. Created by The Finlay Institute and the National Center for Biopreparations (BIOCEN). Originally arose from research at the University of Havana based on a synthetic antigen.
Transformations
In December 2012, the industry transformed as biotechnology companies became part of the BioCubaFarma business group, one of the High-Level Organizations for Enterprise Management (OSDEs) created at that time (today comprised of 32 companies). Biotechnology sector entities were incorporated that previously belonged to the Ministry of Science, Technology and Environment (CITMA) and to the QUIMEFA Business Group, in charge of pharmaceutical production. The group began with establishments in all 15 provinces and was made up of 16 large production companies, eight marketing companies, eleven based abroad and three that offered services.
The integration of the biotechnology and pharmaceutical industries brought benefits for both. BioCubaFarma is a national business entity that includes high-technology companies. Although the integration of both industries could contribute to its success over time, the “company” must be differentiated from the high-tech company.
There are 19 controlled entities abroad, operating either as joint ventures or fully owned by Cuba and its local agencies, or through other modalities. Their products are marketed in more than 50 countries. In some regions they have obtained more than seven hundred licenses.
Biotechnology and the pharmaceutical industry in Cuba are in a position to make a greater contribution to the export portfolio of high-technology products in order to guarantee or recover investment in that area, although the issue with exports is not only a problem of volume, but also of the technological content of what is exported. True technological development always depends on the ability to produce and export new and high-value-added products over time.
However, official Cuban statistics show that exports of chemical and related products—which include medicinal and pharmaceutical products—have declined in recent years, despite the fact that these statistics have not been published since 2016. Biotechnology has slowed the pace of its exports, along with the overall decline in exports. It would be unwise to estimate exports of medicinal and biotechnological products in 2020, given the deep economic crisis brought on by the pandemic.
Statistics show a drop of almost 40% in exports of chemical products and related products between 2015 and 2019. Considering that medicinal and pharmaceutical products make up around 90% of the total exports of chemical products, we could estimate that medicine exports—including, of course, those of biotechnological origin—would be around 240 million pesos annually in 2019. This figure is far below what had been achieved previously. It is not within the established goal range and, therefore, in the future it is unclear what target range should be set.
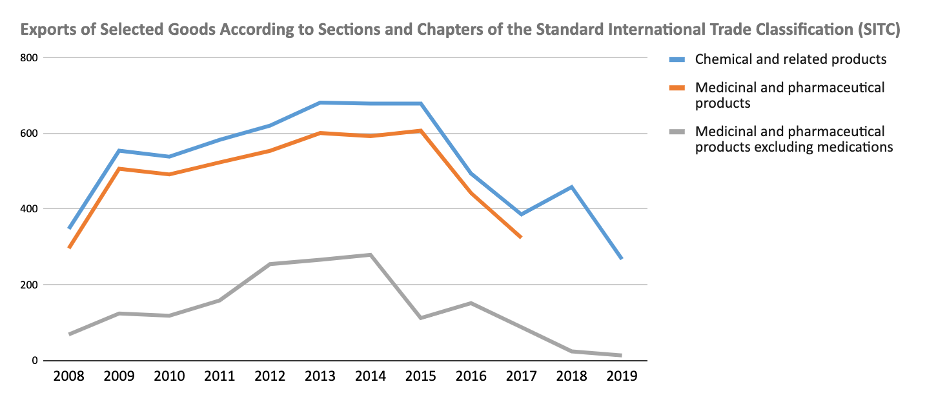
Source: "Anuario Estadístico de Cuba. Sección Sector Externo", Havana.
The advent of BioCubaFarma enabled an influx of foreign capital in this area, especially in the construction of plants and in the Mariel Special Development Zone, where a production and research plant is already being built. Yet in many cases the Cuban State has been reluctant to accept foreign investment in the production or research stages.
Cuban Biotechnology in the Race Against the Greatest Pandemic in a Century
The Cuban biopharmaceutical industry has been key in the strategy to combat the coronavirus pandemic. In the protocols established by the Ministry of Health, most of the products used are domestic and have been effective in seriously and critically ill patients, as well as in certain high-risk groups. In the beginning, the recombinant interferon alfa-2B, already in existence, was used and acceptable results were achieved.
BioCubaFarma is working on 16 projects for new treatments and medical technologies to prevent and treat coronavirus. Four vaccine candidates are at different stages. The Soberana 02 vaccine stands out, and is already being scaled up in large batches for the mass vaccination of the population. The other vaccines are Soberana 01, Abdala, and Mambisa
The Cuban biopharmaceutical industry has been key in combating the coronavirus pandemic. Most of the products are domestic and have been effective in seriously and critically ill patients.
Soberana’s advantage consists of having taken advantage of pre-established platforms, a practice adopted worldwide to shorten timelines in the face of the global impact of the pandemic. This vaccine was based on the RBD protein as the main antigen.
The main Cuban products used during this pandemic have been[i]:
• Biomodulin T. A naturally occurring immunomodulator.
• Hebertrans. A transfer factor.
• Nasalferon. A nasal formulation of recombinant human IFN alpha 2B.
• Hebron. A recombinant human interferon alpha 2B.
• Heberferon R. An IFN alpha-2B + IFN gamma.
• Jusvinza. An immunomodulatory peptide.
• Itolizumab. An anti-CD6 monoclonal antibody.
Itolizumab is being tested in the United States. Two Latin American countries have requested it.
Other drugs are being studied, such as CIGB 300 Peptide, a casein kinase enzyme inhibitor; the VAMENGO-BC meningococcal vaccine; and the CIGB 2020 vaccine. So far, the Cuban immunization scheme includes 13 vaccines. Of these, eight are already produced on the island.
Future Challenges
Cuba has entered an area dominated by some developed countries and a handful of transnational companies. For its competitive development, this sector needs enormous financial resources for research, which Cuba lacks. Hence, the challenge is how to assure its survival in the short term given the country’s conditions. Biotechnology has a high degree of complexity. While it is very profitable when successful, getting there requires investing large sums and a willingness to wait long periods before recouping the investment.
The diversity of destinations with different regulatory environments and the increase in international regulations constitute a barrier to entry for Cuban products in many economies. In each destination country there are regulations to this effect, which requires permanent reinvestment to obtain high standards of quality, a fundamental risk for the Cuban biotechnology sector, especially since the most restricted markets are in developed countries, which have a few monopolistic companies. That is where the greatest benefits can be obtained.
For its competitive development, this sector needs enormous financial resources ... which Cuba lacks.
In general, industry in Cuba has lacked autonomy in making financial decisions. This often makes it impossible to obtain capital when it is needed, and therefore the lack of financial resources is one of its main weaknesses.
Cuba has a low domestic demand for biotech products. In small countries, the domestic market does not provide the scale needed to generate a sufficient demand volume to cover the fixed costs of research and development activities. Therefore, export orientation is essential to achieve economic viability.
In this context, market diversification in the export process becomes a necessity, despite the fact that Cuban biotechnology products are exported today to several countries on all continents. There are still export concentration problems in terms of value, a real limitation for competitiveness.
In the medium term, productivity will depend on the penetration of new markets, but also on creating new products. Thus, domestic companies face the tradeoff between continuing to produce biogenetic products that require less effort (but are less profitable), or making a great effort to renew their product portfolio. This implies greater investment, risk, and profitability in the long term.
An important element of biotechnology is the long duration of the process from research and development to product registration necessary for every biotechnological product, especially medicines. It takes no less than 10 years to develop a biotechnological medicine.
Despite great advances and results, both in the field of science (research) and economics (exports), the biotechnology sector faces major challenges in terms of the concentration of its export destinations and the need for a large injection of financial resources. The country is not in the same position to offer these as it was at the inception of this effort in the 1980s.
Nonetheless, biotechnology is a strategic sector in terms of the multidimensional aspect of Cuba’s development aspirations through 2030. Making it a reality is up to the economic policymakers. To date, Cuban biotechnology has shown excellent results in combating the COVID-19 pandemic, especially in the use of domestic resources and its own vaccines to immunize the entire population.
[i] Source: Granma, August 11, 2020, p.5.

